Fizz-tastic Egg-speriment: Dissolving Eggshells with Vinegar!
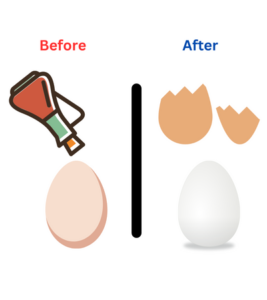
Get ready for some fizz-tastic fun with eggs and vinegar! In this exciting experiment, we’ll discover what happens when we combine vinegar and eggshells. Watch as the vinegar works its magic, dissolving the eggshell and revealing a surprising transformation. Get ready to dive into the world of acids and learn about chemical reactions!
Scientific Principles:
- Acids and Bases: This experiment introduces the concept of acids, such as vinegar, and their ability to react with certain materials. The vinegar contains an acid called acetic acid, which reacts with the calcium carbonate in the eggshell, causing it to dissolve.
- Chemical Reactions: When the vinegar comes into contact with the eggshell, a chemical reaction occurs. This reaction releases carbon dioxide gas, which creates the fizzing effect we observe. Through this experiment, we’ll learn how chemical reactions can cause changes in materials.
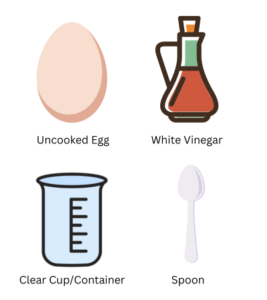
Materials Needed:
- Raw eggs (uncooked)
- White vinegar
- Clear glass or container
- Spoon
- Optional: Paper and pen for recording observations
Equipment Difficulty Rating:
Obtaining the equipment: 2/5 (Eggs and vinegar are easily obtainable from a grocery store.)
Completing the experiment: 2/5 (It’s a simple experiment, but handling the eggs with care is important.)
Steps:
- Get Ready: Find a clean and flat surface to conduct the experiment. Gather all the materials in front of you.
- Place the Egg in the Glass: Carefully place a raw egg into the clear glass or container. Make sure the egg is fully submerged in the vinegar.
- Observe the Egg: Take a moment to observe the egg in the vinegar. What does it look like? What color is the shell?
- Pour in the Vinegar: Slowly pour enough white vinegar into the glass to completely cover the egg. You can use a spoon to help if needed.
- Record Your Observations: If you have paper and a pen, write down what you notice about the egg and its shell before adding the vinegar. This will help you compare your observations later.
- Watch the Fizzing: As soon as you add the vinegar, you’ll see bubbles forming around the egg. This is the reaction between the vinegar and the eggshell. Observe the fizzing and bubbling!
- Let It Sit: Leave the egg in the vinegar for about 24-48 hours. You can cover the glass with plastic wrap to prevent any smells.
- Check on the Egg: After the waiting period, carefully remove the egg from the vinegar using a spoon. Observe any changes to the eggshell’s appearance and texture.
- Record Your Findings: Write down what you observe about the egg and its shell after it has been in the vinegar. How does it look and feel compared to before?
- Explore and Discuss: Discuss what you observed with an adult or a friend. Talk about why you think the eggshell changed and what might have caused it. Have fun learning about the power of vinegar and chemical reactions!
Simple Explanation:
In our Fizz-tastic Egg-speriment, we put an egg in vinegar. The vinegar is a special liquid called an acid. When the egg and vinegar meet, a reaction happens. The acid in the vinegar starts to dissolve the hard shell of the egg. This creates bubbles and fizzing. The shell becomes soft and squishy, revealing the egg inside. Isn’t it amazing how vinegar can change the eggshell?
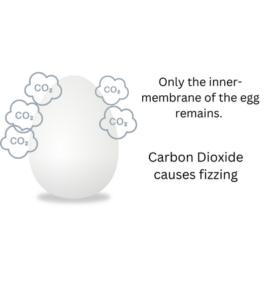
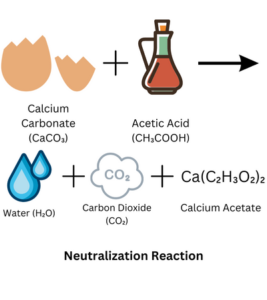
Detailed Explanation:
During the Fizz-tastic Egg-speriment, we explore the reaction between vinegar (which contains acetic acid) and the eggshell, which is made of a substance called calcium carbonate. The acetic acid in the vinegar reacts with the calcium carbonate, causing it to break down and dissolve. This reaction releases carbon dioxide gas, which creates the bubbles and fizzing effect.
The acetic acid reacts with the calcium carbonate in a chemical process called neutralization. As the acid dissolves the calcium carbonate, it forms new substances, such as water and a compound called calcium acetate. The release of carbon dioxide gas is what we observe as the bubbles and fizzing.
As the eggshell dissolves, it leaves behind the egg’s inner membrane, which is more flexible and allows us to gently hold the egg without it breaking. The reaction between the vinegar and the eggshell helps us understand the properties of acids, the concept of neutralization, and how chemical reactions can cause changes in materials. It’s a captivating exploration of chemistry and the power of vinegar!
Optional Follow-on Experiment: Exploring Osmosis with Eggs and Saltwater
Description:
After the Fizz-tastic Egg-speriment, let’s dive into another fascinating scientific concept called osmosis. In this experiment, we’ll investigate how osmosis works by placing our eggs in saltwater. Get ready to observe the effects of water movement through a semi-permeable membrane and discover how it can cause changes in the egg!
Scientific Principle:
- Osmosis: This experiment introduces the concept of osmosis, which is the movement of water molecules across a semi-permeable membrane from an area of higher water concentration to an area of lower water concentration. By placing the egg in saltwater, we’ll explore how osmosis can cause water to move in or out of the egg, leading to changes in its size and texture.
Materials Needed:
- Hard-boiled eggs (shells removed)
- Salt
- Clear glass or container
- Water
- Measuring spoons
- Optional: Paper and pen for recording observations
Steps:
- Get Ready: Gather all the materials and find a clean and flat surface to conduct the experiment.
- Prepare the Saltwater Solution: In a clear glass or container, pour 2 cups of water. Add 2 tablespoons of salt to the water and stir until the salt is dissolved. This creates the saltwater solution.
- Place the Egg in Saltwater: Carefully place a hard-boiled egg into the saltwater solution. Ensure the egg is fully submerged in the solution.
- Observe and Record: Take a moment to observe and note down the egg’s appearance and texture before placing it in the saltwater. This will help you compare your observations later.
- Let It Sit: Leave the egg in the saltwater for about 24-48 hours. You can cover the container with plastic wrap to prevent any smells.
- Check on the Egg: After the waiting period, carefully remove the egg from the saltwater solution. Observe any changes to the egg’s appearance and texture.
- Record Your Findings: Write down what you observe about the egg after it has been in the saltwater solution. How does it look and feel compared to before?
- Explore and Discuss: Discuss your observations with an adult or a friend. Talk about why you think the egg changed and what might have caused it. Reflect on the concept of osmosis and how water moves through the egg’s membrane.
Remember, the eggs used in this experiment are hard-boiled and do not have shells. Avoid tasting or consuming the saltwater or the egg. Enjoy exploring the intriguing world of osmosis and the effects it can have on materials like eggs!
Note: Adult supervision is recommended, especially when handling glass containers and saltwater.

