Slime Time: Create Your Own Colorful, Stretchy Slime!
Description:
Get ready for a slimy adventure! In this fun and hands-on experiment, you’ll become a scientist and create your very own homemade slime. Discover the fascinating properties of slime and explore how different ingredients interact to form this ooey-gooey substance. Let’s dive into the world of polymers and have a blast with colorful, stretchy slime!
Scientific Principles:
- Polymers: This experiment introduces the concept of polymers, which are long chains of molecules. Slime is made up of polymers that have a unique ability to stretch and flow. You’ll explore how the ingredients in slime interact and create these fascinating properties.
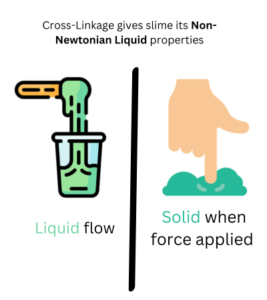
- Non-Newtonian Fluids: Slime is an example of a non-Newtonian fluid, which means it doesn’t follow the typical rules of liquid or solid behavior. It can behave like a liquid or a solid depending on the forces applied to it. You’ll observe how slime can flow like a liquid when gently handled and act like a solid when you apply force to it.

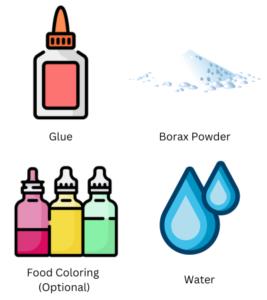
Materials Needed:
- White school glue
- Borax powder (available at hardware stores or online)
- Water
- Food coloring or liquid watercolors (optional)
- Mixing bowl
- Measuring cups and spoons
- Spoon or popsicle stick for stirring
- Sealable plastic bags or airtight containers for storage
Equipment Difficulty Rating:
Obtaining the equipment: 3/5 (Most materials can be easily found at home or purchased at a nearby store.)
Completing the experiment: 2/5 (It’s a straightforward experiment, but adult assistance may be needed when handling borax powder.)
Note: Adult supervision is recommended, especially when handling borax powder. Avoid contact with eyes or ingestion of any ingredients.
Steps:
- Get Ready: Find a clean and flat surface to conduct the experiment. Gather all the materials in front of you.
- Measure the Glue: Pour 1/2 cup of white school glue into a mixing bowl. This will be the base of our slime.
- Prepare the Borax Solution: In a separate container, mix 1 cup of water with 1 teaspoon of borax powder. Stir until the borax powder is dissolved. This is the special solution that will transform the glue into slime.
- Optional: Add Color: If you want colored slime, add a few drops of food coloring or liquid watercolors to the glue mixture. Stir well until the color is evenly distributed.
- Combine the Ingredients: Slowly pour the borax solution into the glue mixture, stirring continuously. You’ll notice the mixture thickening and coming together.
- Stir, Stir, Stir: Keep stirring until the mixture becomes too thick to stir with a spoon or popsicle stick. At this point, you can use your hands to knead and mix the slime together.
- Explore and Play: Take the slime out of the bowl and start playing with it! Stretch it, squeeze it, and see how it flows. Observe its unique texture and the way it behaves.
- Store for Later: When you’re finished playing, store the slime in a sealable plastic bag or an airtight container to keep it fresh for future fun.
Remember, if the slime feels sticky, you can add a little more borax solution or water to adjust the consistency. Enjoy your homemade slime and have a squishy, sensory-filled adventure!
Note: Adult supervision is recommended, especially when handling borax powder. Avoid contact with eyes or ingestion of any ingredients. Wash hands thoroughly after playing with the slime.
Simple Explanation:
In our Slime Time experiment, we mix glue and a special solution made from borax powder and water. When we combine these ingredients, they go through a chemical reaction called cross-linking. This reaction creates long chains of molecules called polymers. These polymers give the slime its stretchy and squishy properties. So, when we play with the slime, we’re exploring the world of polymers and having a gooey and fun time!
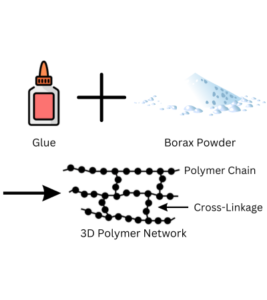
Detailed Explanation:
During the Slime Time experiment, we delve into the science of polymers and chemical reactions. The glue we use contains molecules called polymers. These are like long chains made up of smaller building blocks. When we add the borax solution to the glue, a process called cross-linking occurs. The borax molecules link the polymer chains together, creating a three-dimensional network of interconnected polymers.
As a result of cross-linking, the slime changes from a liquid (the glue) to a non-Newtonian fluid (the slime). Non-Newtonian fluids have unique properties because their flow can change depending on the force applied to them. Slime acts like a liquid when we let it flow slowly, but when we apply force, like squeezing or stretching, it behaves like a solid.
The cross-linked polymers in the slime give it its stretchy, gooey, and squishy nature. When we play with the slime, we’re exploring how the polymers interact and respond to different forces. It’s a fascinating example of the science behind materials and chemical reactions.

